How to Identify Optimal Materials for Biopharma Single-use Solutions
So, you’re ready to assemble your biopharma process for advanced therapy manufacturing.
But have you considered how the materials used to construct the single-use bags, tubing, transfer sets, and manifolds used in therapy manufacturing will benefit your processes?

Explore material considerations for 7 steps of the advanced therapy manufacturing process.
1
Cell Culture
Choose bioinert and gas-permeable materials such as a breathable polyolefin to create the ideal conditions for metabolism and cell viability.
2
Transfer
Efficiently transfer critical cell-based fluids by leveraging durable yet flexible polyethylene or polyvinyl chloride bag materials that engage critical gas transmission properties during fluid transfer.
Enable reliable, seamless connectivity and aseptic fluid transfer while minimizing cell shear and optimizing fluid dynamics with di(2-ethylhexyl) phthalate (DEHP)-free polyvinyl chloride, silcone, or thermoplastic elastomer tubing.
3
Formulation
Select materials made of low-extractable polyolefin to ensure purity during mixing and connectivity with large volume or batch processing – and understand how they interact with formulation additives such as dimethyl sulfoxide (DMSO).
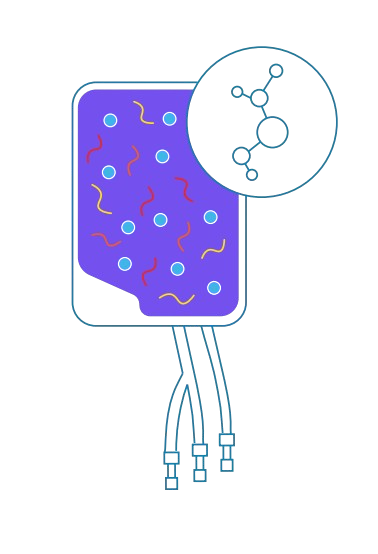
4
Storage
Long-term storage using layered polyethylene films provides durability, impermeability, and a critical gas barrier. For high-volume applications, multi-layer materials can provide combined properties for strength and flexibility.
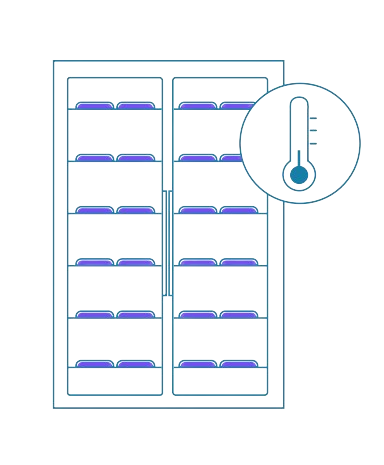
5
Filtration
Combine or fine-tune polymer-based textile components constructed of materials such as polyester or nylon to optimize filtration and durability under pressure or degradation conditions.
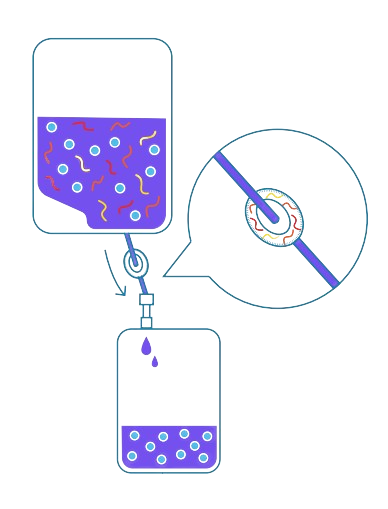
6
Waste Disposal
Capture process waste using materials such as polyethylene and polyvinyl chloride that balance durability and cost-effectiveness while operating under fluctuating temperatures and pH levels.
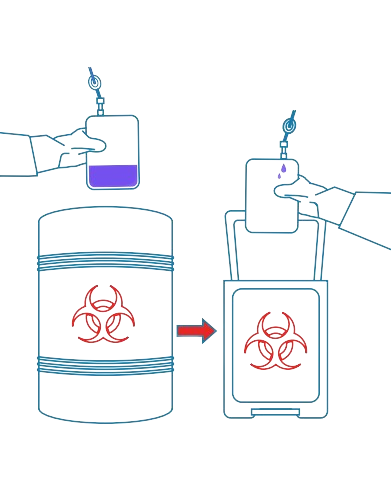
7
Cryopreservation
Harness the benefits of flexible, durable polymers like thermoplastic elastomers and low-temperature polyolefin to prevent cold cracking, ultimately safeguarding the end product for therapy administration.